Evaporating Multi-Component Droplets
Sessile droplets typically evaporate faster near their edge than at their center. In droplets that consist of only one single liquid, capillarity compensates these losses, causing a strong flow toward the edge of the droplet. This is the reason why coffee-stains look like rings: coffee particles move with the capillary flow and are deposited in a ring around the droplet. If, however, a solute or a second, less volatile liquid is present in the droplet, this component will enrich near the edge of the droplet. In contrast to inert particles, however, liquids or solutes influence surface tension, which is why their local enrichment causes another type of flow, known as Marangoni convection. It can be directed toward the contact line, or away from it, depending whether the residual component increases or decreases the surface tension, respectively. Both cases are equally important, and applications range from printing to cleaning. We work on a quantitative understanding and try to reveal underlying universal concepts and scaling laws that allow to predict the behavior of droplets in technological applications.
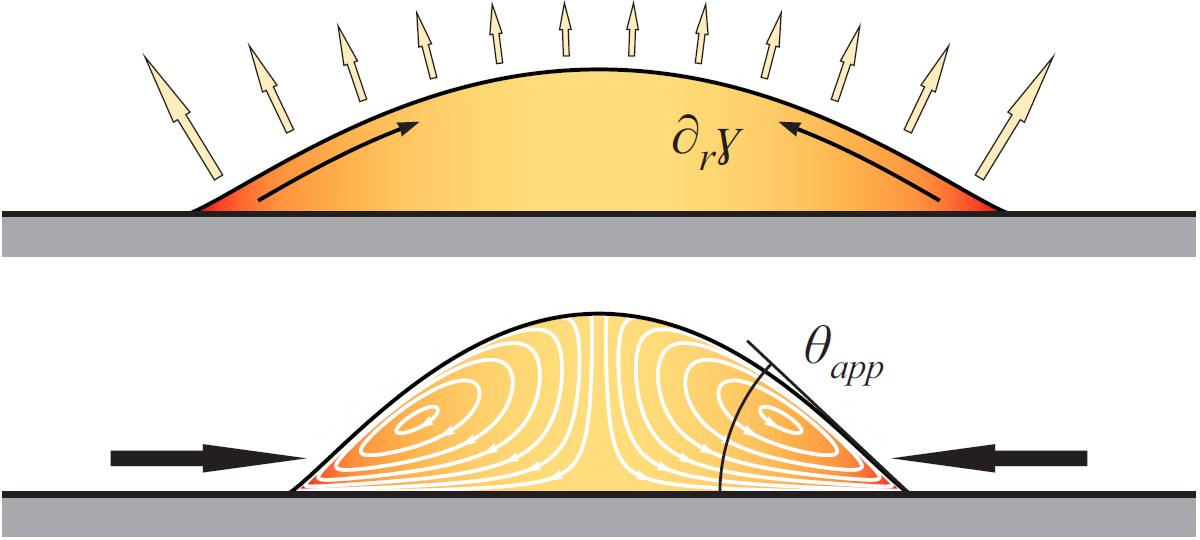
If this flow is directed the other way round, e.g. as for glycerol/water mixtures, and if the pure liquids wet the substrate, these flows even dominate the shape of the droplet: instead of spreading and covering the whole substrate, the liquids contract into a droplet with a stationary non-zero contact angle, as if the substrate would be non-wetting [Karpitschka et al., Langmuir 33 (2017) 4682]. Why is this important? Virtually any micro-electronic circuit, as in phones or computers, is made by wet processing and has to be dried at the end. So far, the wetting behavior of liquids on solids was known to be determined by the material’s surface energies (that’s Young’s law) and surface topography (Wenzel’s law), and nothing much could be changed about this. Drying microelectronics was (and is) a challenge, because material and topography are prescribed by the requirements of the electronics, not the drying; droplets would simply stick to the circuit. However, these contracted droplets seemingly forget about Young’s and Wenzel’s laws. Instead, their wetting behavior is governed by evaporation and can be controlled remotely by changing the atmospheric composition around the droplet [Karpitschka et al., Langmuir 33 (2017) 4682].
One may think that this effect can be enhanced simply by using more surface active components i.e., materials that reduce the tension of a surface by adsorbing to it. However, such materials frequently also adsorb to solid surfaces, causing another effect that changes the wetting behavior: autophobing. Adsorption to a solid forces the molecules into an orientation in which they expose their ‘tails’ that repel liquids. Even if the liquid is made of the very same molecules (simply because of the molecular orientation). Recently, we could show the coexistence of Marangoni contraction and autophobing, depending on ambient conditions [Hack et al., Langmuir 37 (2021) 3605]
Have you ever enjoyed a glass of Ouzo (or Pastis, or Absinthe, or Pernod, or ...), adding some fresh water, noticing that it turns opaque? Liquids do not always mix in all proportions: oil would not blend into water, but rather form a thin film at the surface. What now, if this occurs while a droplet, like the ones we just discussed, sits on a nice and clean surface? The process of phase separation would occur first in places where the composition changes most, that is at the contact line, exactly where all the Marangoni flow happens. In our study [Chao et al., PNAS (2022)], we could show that phase separation enhances the spreading motion, and that this occurs before any microdroplets become visible inside the main droplet, presumably because van der Waals interactions change the equilibrium conditions at the contact line.
