Interfacial Singularities
What is a singularity? If we describe a physical phenomenon with a continuum theory, like the adhesion of a flat elastic material to a rigid sphere, we frequently observe that a physical quantity, in this case the stress in the solid, becomes infinitely large at some “singular point”. Here the singular point is the edge of the contact. In nature however, nothing becomes truly infinite, so there must be something wrong with the continuum theory. Obviously, the equations did not take into account the discrete (atomic) nature of materials. But fully describing all atoms is a formidable task, because there’s so many of them – how could we “save” continuum theory, and take microscopic physics into account? Here are a few recent discoveries that illustrate our research in this field.

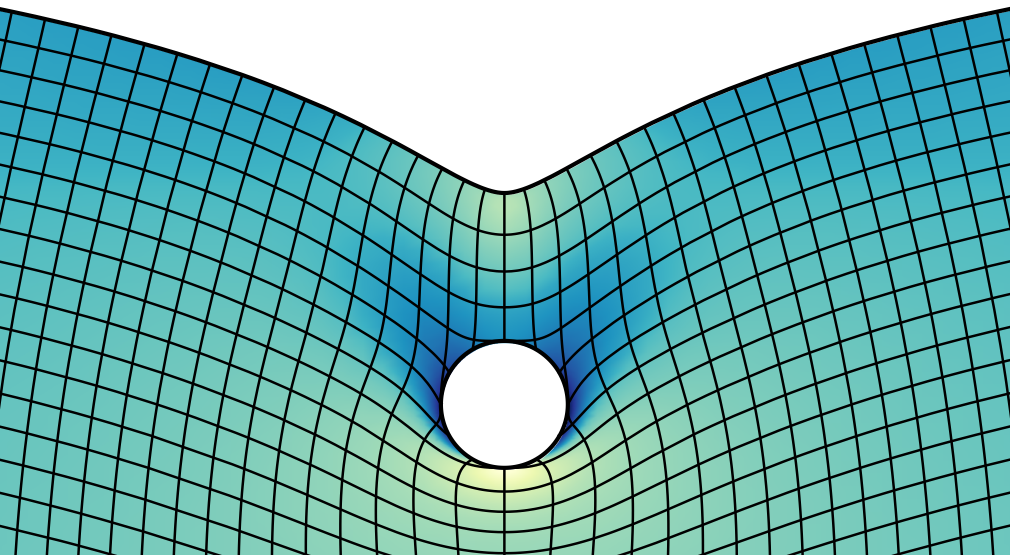
The concept of surface tension is one example: molecules in the bulk of a liquid interact with each other on all sides. The molecules at the interface to a gas are missing half of their interaction partners. This causes surface tension, which can be included in continuum theories. Scientists agree that surface tension is utterly important to describe liquids. For solids, surface tension was largely ignored in the past because there, the consequences are much smaller due to the rigidity of a solid. But approaching a singularity, it becomes important again. The distance below which solid surface tension matters can be calculated by comparing surface tension to stiffness, ɣ/E. This gives a length, the elastocapillary length. Within this range, the edge of an elastic contact is shaped by solid capillarity. After a couple of lines of variational calculus, one can show that surface tension also provides a boundary condition for the angle of contact between the elastic material and the sphere [Karpitschka et al., Soft Matter 12 (2016) 4463]. This is equivalent to what is known for liquids as Young’s contact angle. On small scales, adhesion and wetting are equivalent!
Compressing a slice of soft bread, one can observe an inward fold with a self-contacting surface of the bread. A very similar effect is responsible for the morphology of our brains, where an inward fold is termed “sulcus”. Sulci form because the outer part of the brain grows faster than the inner part. Despite its importance in biology and engineering, the morphology of such inward-folds, in mechanics called creases, remained unexplored. In our paper [Karpitschka et al., Phys. Rev. Lett. 119 (2017) 198001] we discovered a continuous precursor to a crease, a “furrow”: it can be generated by deforming a soft surface inward, for instance by deflating a liquid inclusion beneath the surface, or pulling a solid inclusion downward. The furrow bifurcates sub-critically into a crease, when the surface region becomes compressed beyond a critical point: The free surface folds upward into a self-contact. In the reverse direction, unfolding proceeds smoothly. Both, the furrow and the crease, are described by a simple mathematical concept: self-intersecting curves. The “simplest” set of curves that undergo self-intersection belongs to the universality class of “cusps”. When the radius of the surface reaches zero, it forms an interfacial singularity and folds. Of course, for a solid, self-intersection is replaced by a self-contact.
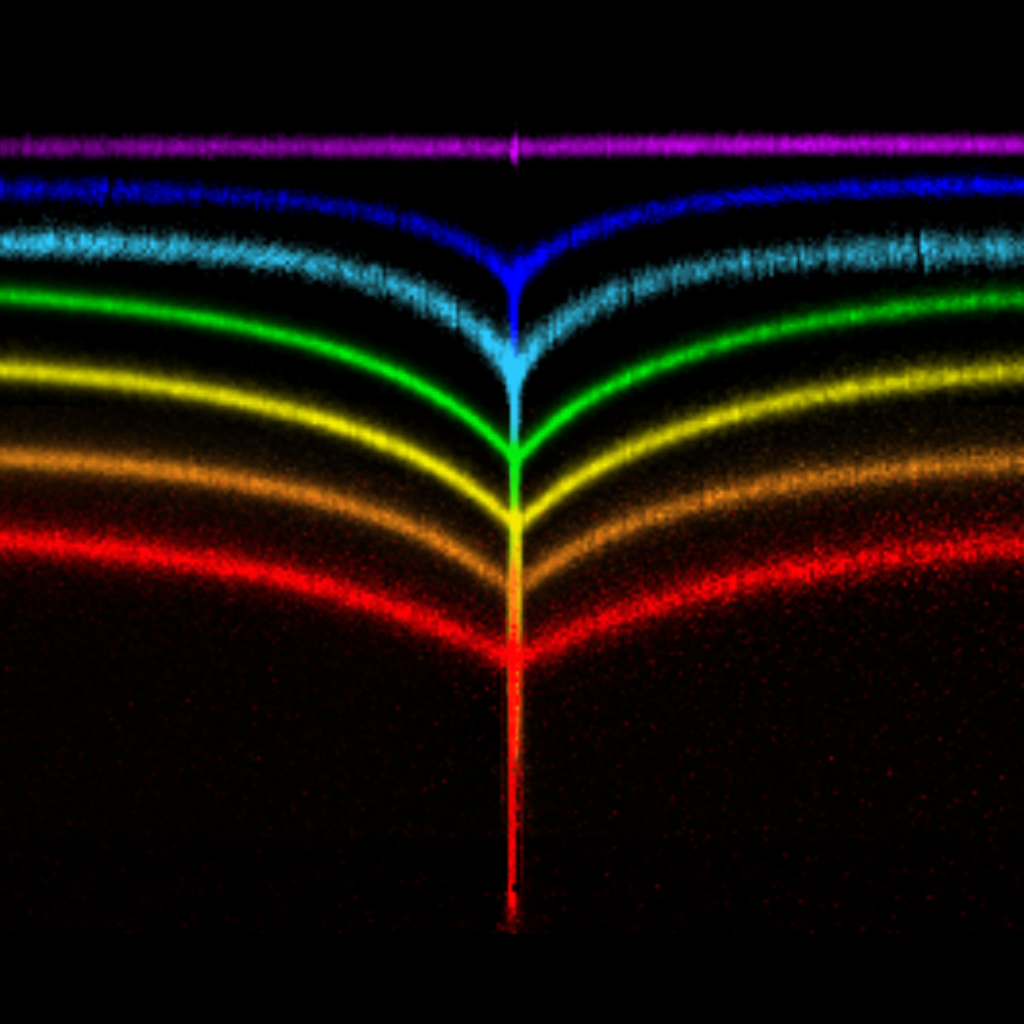
But what happens if the material is tacky? Tackiness is a consequence of compliance and adhesion i.e., that the material reduces its surface energy by snugly fitting onto the contacting body. In a “sticky crease”, this happens to a self-contacting surface. Surprisingly, we found that the micro-morphology at the edge of a self-contact changes drastically if the surface is tacky [van Limbeek et al., Phys. Rev. Lett. 127 (2021) 028001]: Instead of a gentle touch-down, we find a T-shape, with a sharp fold on either side of the opening contact. This is caused by surface tension, which is fully eliminated in the seemingly perfect contact, leaving no other option to balance forces. Similar to the contact between an elastic material and a sphere (see above), this “inner region” is dominated by solid capillarity, making it similar to the wetting of a liquid on a solid surface. We could even recover the typical behavior of liquids that spread or recede on imperfect surfaces, but this time, for a closing or opening self-contact: The contact edge is subject to contact line pinning, meaning that the history of the contact matters: Creases look different during folding and unfolding.
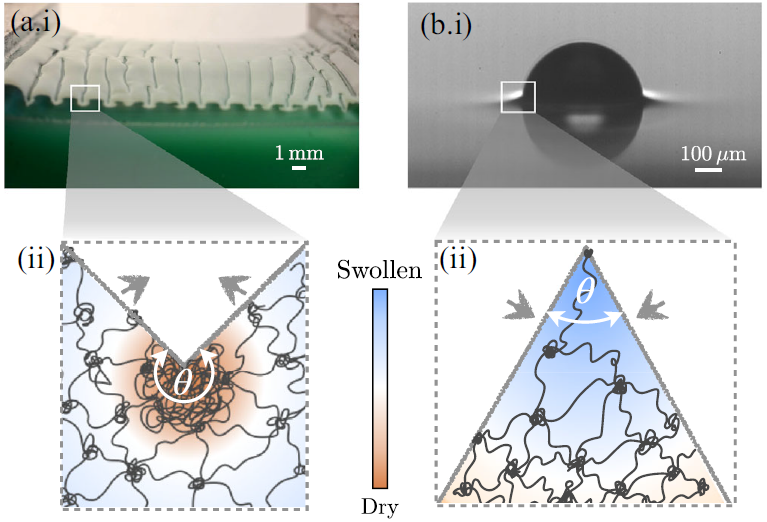
Most real-world soft materials are actually a combination of a fluid, dissolved in an elastic network: This is called a gel. Gels act like sponges: they can soak up more of the liquid they like (dunk a gummi bear in a glass of water over night and see what happens!) or expell it when squeezed. The folds we just discussed introduce a huge (diverging, that is, becoming infinite when approaching the tip) pressure i.e., should squeeze out or soak up (depending on the sign) fluid. We managed to compute how the liquid is actually supposed to be distributed around such folds [Flapper et al., Phys. Rev. Lett. (2023)]: Folding up, as in wetting ridges, liquid accumulates near the tip of the fold, whereas for creases, the tip dries out!
